In silico POC for ChemPass Derivatization Design technology
Reference Donepezil Study
Generative design and structure evolution by structural transformations have gained significant popularity in the design of new chemical classes or analogs in lead optimization. These methods can generate new molecules for in silico evaluation that are chemically mostly reasonable. Synthetic feasibility is not incorporated in the design methods thus all designed molecules will ultimately require either manual verification of synthetic pathways or computational retrosynthetic analysis. Furthermore, it has been reported that it can be challenging to create molecules that retain high probability of fulfilling both the design criteria as well as sufficient novelty for medicinal chemists.
A seminal paper in Nature, 492, pp 215–220 (2012), “Automated design of ligands to polypharmacological profiles” by Hopkins et al. demonstrated how such automated design can contribute to rapid and efficient lead optimization when coupled with active learning using ML or deep learning models. The authors describe the design and validation of novel molecules with different pharmacological profiles (Table 1).
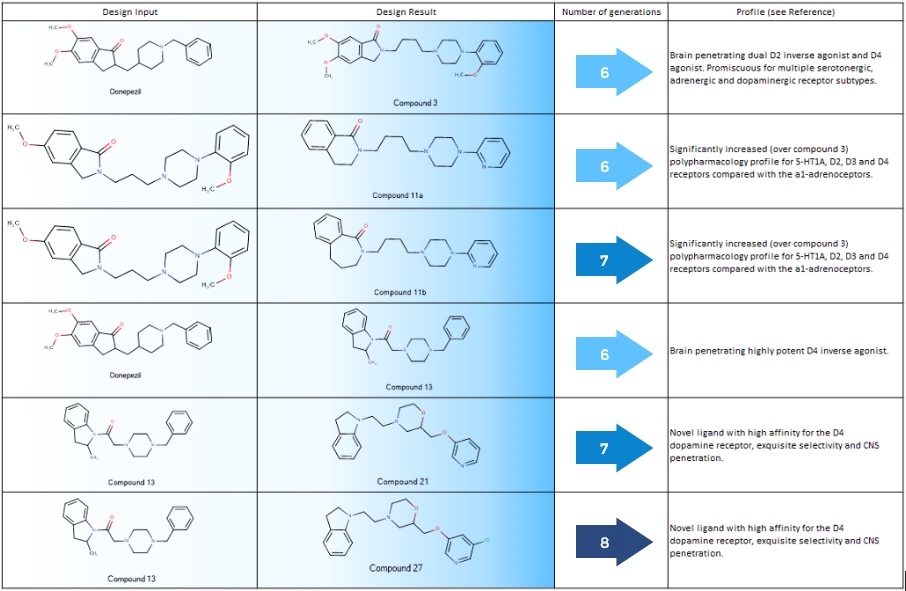
Selected new molecules generated via the generative design method from their respective starting molecules via 6-8 design generations.
Chempass Comparative Donepezil Study
ChemPass has developed a Derivatization Design technology for its AI-assisted lead optimization platform that utilizes rule-based AI-enabled in silico forward synthesis. The technique evaluates multiple synthetic routes and relevant related reagents to synthetically feasible analogs of leads that have the potential of project relevant pharmacological profiles. Four different analyses are utilized to assess the relevance of the new chemical space for the project and naturally all designed molecules carry detailed synthetic route, reagent cost and other relevant information for decision making.
We have applied our Derivatization Design method for the 6 new compounds in Table 1. All designs were initiated from Donezepil (unlike in the above article where several molecules were created via new cycles starting from virtually designed inputs). As seen in Figure 1 below, 3 new molecules could be created in 2 generations, 1 in 3 generations, and 2 molecules needed 6 generations from Donezepil.
Importantly, all 6 notable structures identified in the article could be accessed by Derivatization Design and in all cases in fewer optimization cycles than those reported in the paper for generative design.
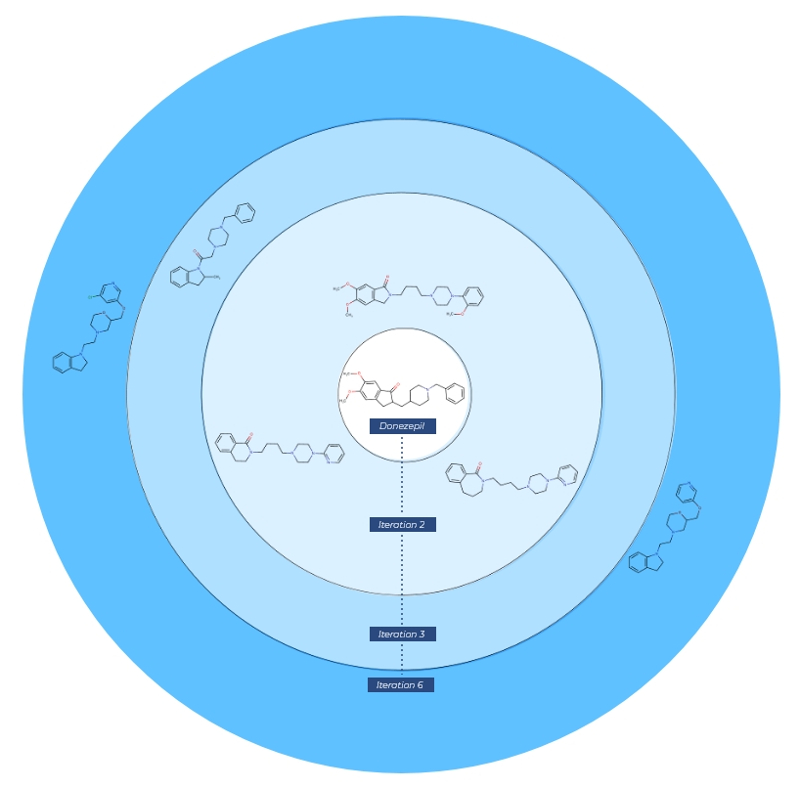
ChemPass Derivatization Design results for the 6 reported molecules with different pharmacological profiles.